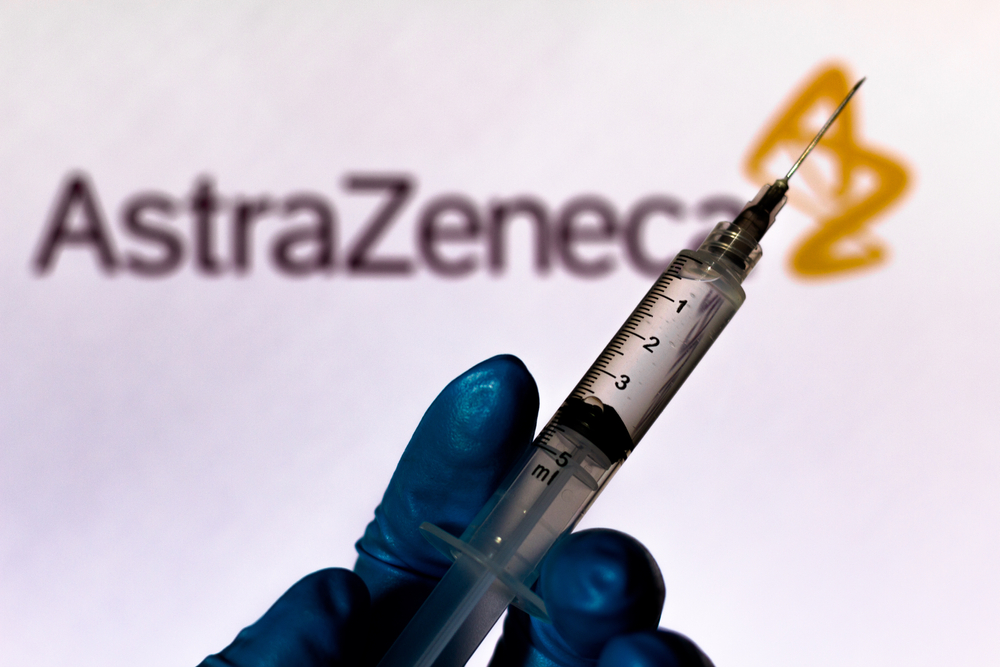
A Brazilian man taking part in clinical trials of the Covid-19 vaccine developed by Oxford University and British-Swedish pharmaceutical firm AstraZeneca has died, according to Brazil’s National Sanitary Surveillance Agency (Anvisa).
In a statement issued today, Anvisa reported that the volunteer died last Thursday, and the agency was informed on Monday.
In a statement, Oxford University declared that “following careful assessment of this case in Brazil, there have been no concerns about the safety of the clinical trial and the independent review in addition to the Brazilian regulator have recommended that the trial should continue.”
In addition, both Bloomberg and Brazilian newspaper O Globo cite unidentified sources linked to the trial who confirmed that the patient did indeed receive the placebo.
The late-stage Oxford vaccine trial had already been interrupted in September, when a patient in the UK contracted an unexplained illness, suspected to be a rare inflammatory disorder known as transverse myelitis. Trials resumed this month, when it was concluded that the illness was not connected to the vaccine.
The deceased volunteer was a 28-year-old doctor who worked on the frontline treating Covid-19 patients in Rio de Janeiro.
The Oxford-AstraZeneca vaccine has been involved in a Brazilian political feud, with President Jair Bolsonaro focusing his government’s efforts on purchasing the British-made vaccine, to the detriment of the CoronaVac — developed by Chinese firm Sinovac — which has shown more promising results.
CoronaVac is backed by São Paulo Governor João Doria, who has promised to immunize the state’s population at the beginning of next year. President Bolsonaro sees Mr. Doria as his main political rival on the Brazilian right-wing, and believes the success of CoronaVac would translate into a political win for Mr. Doria.
 Support this coverage →
Support this coverage →
 Search
Search