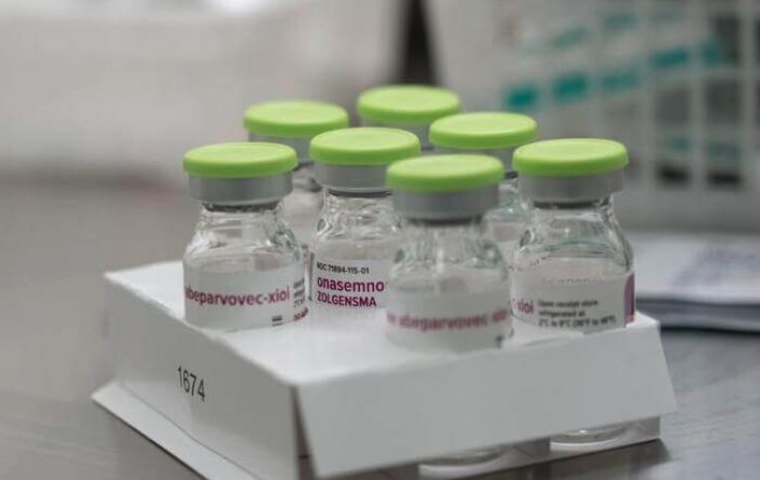
President Jair Bolsonaro has moved to zero import tax rates on gene therapy Zolgensma, used to treat spinal muscular atrophy (SMA) in children. The drug has often been tipped as the ‘most expensive in the world,’ due to its current cost of over USD 2 million for the one-dose treatment.
Crucially, however, Zolgensma has not even been cleared for sale by Brazil’s sanitary regulator Anvisa, and former members of the agency and the Health Ministry told newspaper Folha de S.Paulo that this could be opening a dangerous precedent.
“Until now, no drugs unlicensed by Anvisa have been supplied by the courts, only in exceptional cases which are treated as anomalies,” said Hillegonda Maria Novaes, a professor from the medicine faculty of the University of São Paulo. However, experts do not see a particular risk with Zolgensma, as it has been authorized by leading pharmaceutical regulators around the world, such as the Food and Drug Administration in the U.S., and the EU’s European Medicines Agency.
Another fear is that this government endorsement will lead to an uptick in litigation from patients’ families to attempt to obtain the drug for free via Brazil’s public health service. The extremely high cost of the treatment would make such rulings exceedingly cumbersome for the national system.
Indeed, while the gene therapy is already prohibitively expensive anywhere in the world, Brazil’s tax rules make the cost of Zolgensma even more astronomical. On top of the USD 2 million price tag, buyers would be subject to a 4 percent import tax — an extra USD 80,000, now scrapped by the government — and around USD 360,000 in state goods and services tax.
 Support this coverage →
Support this coverage →
 Search
Search






































